Two lots of blood pressure medicine, Quinapril and Hydrochlorothiazide tablets have been recalled because they contain too-high levels of nitrosamines.
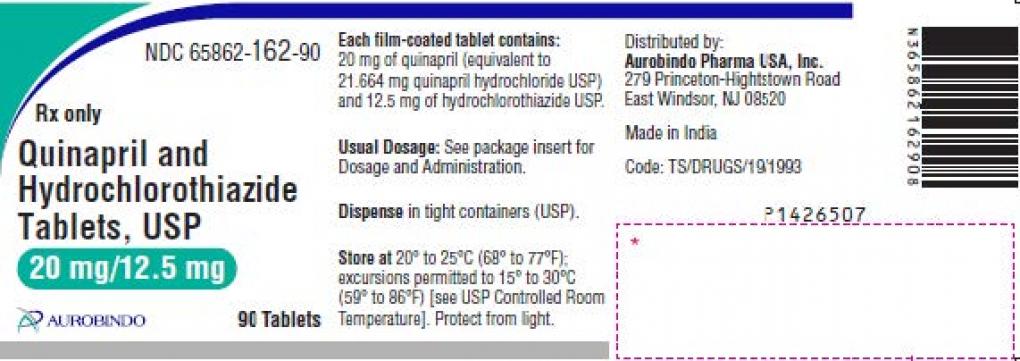
Aurobindo Pharma USA, Inc. issues the voluntary recall of two (2) lots (refer table below) of Quinapril and Hydrochlorothiazide Tablets USP 20mg / 12.5mg, to the consumer level from the US market due to presence of Nitrosamine Drug Substance Related Impurity (NDSRI), N-Nitroso-Quinapril above the proposed interim limit.
Aurobindo Pharma USA, Inc. began shipping of the subject batches, QE2021005-A and QE2021010-A to customers nationwide May 2021.
| NDC No. | Product Name, strength, and pack | Lot number | Expiry |
|---|---|---|---|
| 65862-162-90 | Quinapril and Hydrochlorothiazide Tablets USP, 20mg / 12.5mg, 90’s HDPE bottle |
QE2021005-A | 01/2023 |
| QE2021010-A |
Risk Statement: Nitrosamines are common in water and foods, including cured and grilled meats, dairy products and vegetables. Everyone is exposed to some level of nitrosamines. These impurities may increase the risk of cancer if people are exposed to them above acceptable levels over long periods of time. To date, Aurobindo Pharma USA, Inc. has not received any reports of adverse events related to this recall.
Quinapril and Hydrochlorothiazide Tablets, USP are fixed-combination tablet that combines an angiotensin-converting enzyme (ACE) inhibitor, quinapril hydrochloride, and a thiazide diuretic, hydrochlorothiazide. This product is indicated for the treatment of hypertension, to lower blood pressure. Patients should contact their doctor or health care provider about whether to continue taking their medication, or whether to consider an alternative treatment prior to returning their medication.
Quinapril and Hydrochlorothiazide Tablets USP 20 mg / 12.5 mg are “Pink colored, scored, round shaped, biconvex, film-coated tablets, debossed with ‘D’ on scored side and ‘19’on other side”, supplied in 90’s HDPE bottle.
For more on the recall, click here